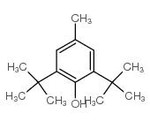
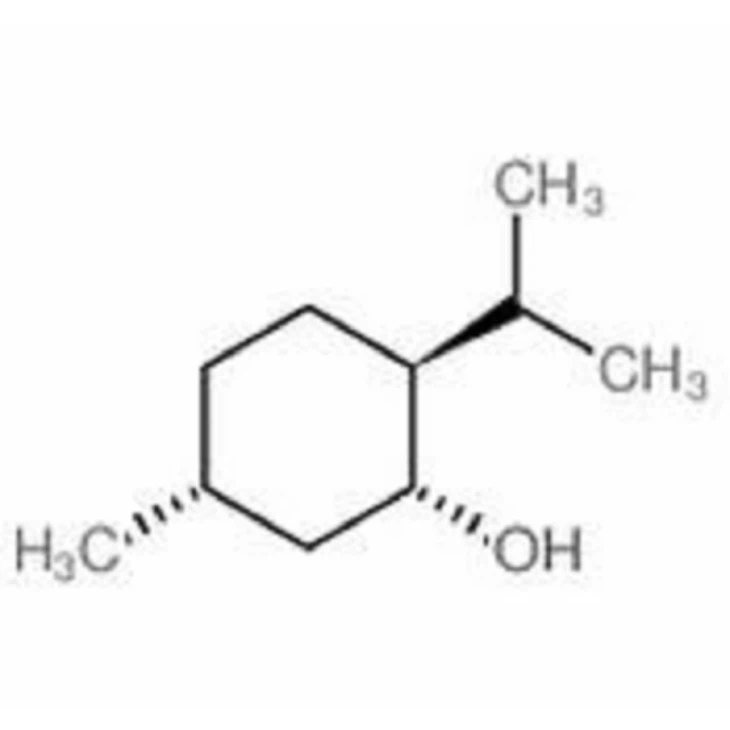
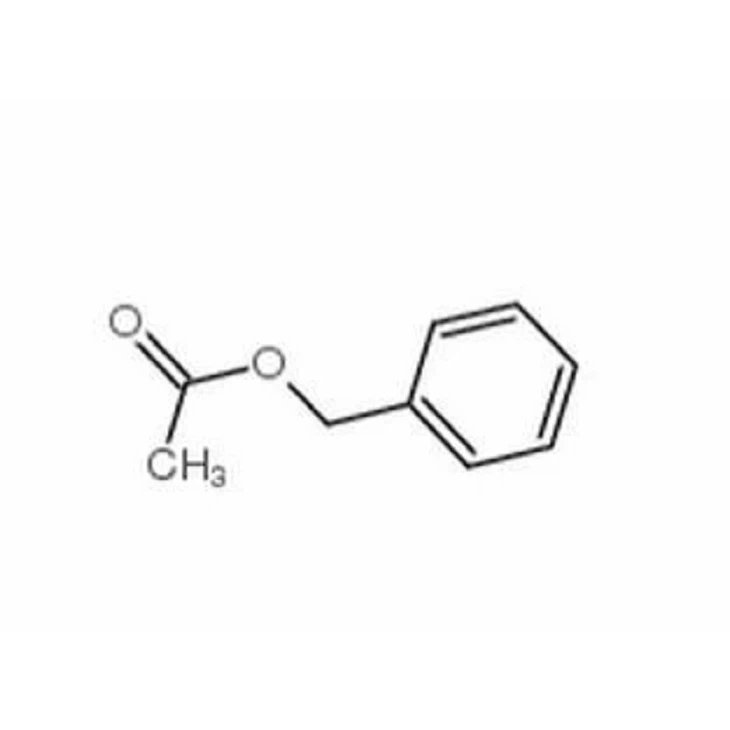
Diphenylmethane
Why Choose Us?
Rich Experience
HANGZHOU BETTER CHEMTECH LTD is a comprehensive chemical company specializing in Chemical Production, Management, Application Development and Brand Promotion. BETTER CHEMTECH currently has two independent sub-brands: Chemintel and Eubiotics.
Professional Team
Our company has a team of highly experienced professionals in both China and India. Under the environmental protection problems faced by the industry, we strive to ensure a stable supply of products, and provide customers with the best supply chain solutions.
Quality Control
As a reliable supplier with many years of professional experiences in the chemical industry, product quality is our primary consideration. We will provide sample testing and confirm the specifications to ensure your satisfactory of the finally shipment goods.
Application of the Product
Our products have application in the fields of pharmaceuticals & fine chemicals, pesticides, dyes, flavors and fragrances, feed additives, green fibers & others.
Diphenylmethane is an organic compound with the formula (C6H5)2CH2 (often abbreviated CH2Ph2). The compound consists of methane wherein two hydrogen atoms are replaced by two phenyl groups. It is a white solid.
Diphenylmethane is a common skeleton in organic chemistry. The diphenylmethyl group is also known as benzhydryl.
L-Menthol 2216-51-5 CAS NO: 2216-51-5 HS CODE: Formula: C10H20O Molecular Weight: 156.26500.
Benzyl acetate is an organic compound. Benzyl acetate is found naturally in many flowers. It is the primary constituent of the essential oils from the flowers jasmine, ylang-ylang and tobira
The Medicinal Use of Diphenylmethane
In the synthesis of luminogens, diphenylmethane plays an important role for aggregation-induced emission (AIE). In the science of luminogenic synthesis, diphenylmethane is widely used to make luminogens. Diphenylmethyl potassium (DPMK) is a polymerization initiator made from this compound. Polymerization agents containing diphenylmethyl potassium. The fragrance industry uses diphenylmethane both as a fixative and in soaps to scent them. This substance can be used in pesticides and insecticides to synergize with pyrethrin. The plasticizer diphenylmethane can be used as a dye carrier, a solvent for dyes, or a plasticizer to improve dyeing properties. Diphenylmethane has improved the thermal and lubricating properties of saturated linear polyesters as well as jet fuels by adding it to them.

Applications of Diphenylmethane
● Diphenylmethane, is used as an adhesive chemical composition for making flexible laminates for food packaging.
● Diphenylmethane is used as a fixative and as a fragrance in soaps
● This is used in insecticides and pesticides to synergize with pyrethrin
Diphenylmethane (C13H12) is a white solid substance that is soluble in alcohol and insoluble in water. Hence , at room temperature , Diphenylmethane is a solid. It has a melting point of 26.5-27.5 °C and a boiling point of 256 °C.
Diphenylmethane is used in the manufacturing of plastics, particularly polycarbonate and polyurethane. It is also used as a starting material for the preparation of dyes and fragrances, as well as in the production of resins and inks.
The organic compound diphenylmethane has the chemical formula (C6H5)2CH2. The substance is made up of methane with two phenyl groups in lieu of the two hydrogen atoms. It is completely white.In organic chemistry, it is a typical skeleton. The term "benzhydryl" also applies to the diphenylmethyl group.It is made through Friedel-Crafts alkylation of benzyl chloride with benzene when a Lewis acid, such as aluminium chloride , is present.
Diphenylmethane is used in the fragrance business as a fixative and to smell products. This chemical can work in conjunction with pyrethrin in pesticides and insecticides. The dye carrier, dye solvent, or plasticizer diphenylmethane can all be utilised to enhance the qualities of dyeing.
What Functional Groups Are in Diphenylmethane
A diarylmethane that is methane substituted by two phenyl groups. Diphenylmethane is an organic compound with the formula (C6H5)2CH2 (often abbreviated CH2Ph2). The compound consists of methane wherein two hydrogen atoms are replaced by two phenyl groups.
How Is Diphenylmethane Formed
Diphenylmethane is an organic compound with the formula (C6H5)2CH2 (often abbreviated CH2Ph2). The compound consists of methane wherein two hydrogen atoms are replaced by two phenyl groups. Diphenylmethane is a common skeleton in organic chemistry.
Diphenylmethane can be prepared from benzene and benzyl chloride with aluminum chloride, hydrogen fluoride, beryllium chloride, the double salt of aluminum and sodium chlorides, zinc dust, zinc chloride, or aluminum amalgam as a condensing agent. In another method, benzene and benzyl alcohol furnish diphenylmethane on treatment with boron fluoride, hydrogen fluoride, or beryllium chloride. Diphenylmethane has also been prepared from benzene, methylene chloride, and aluminum chloride, and from benzene, formaldehyde, ethanol, and concentrated sulfuric acid. The reduction of benzophenone to diphenylmethane has been affected by hydriodic acid and phosphorus, sodium and alcohol, and fusion with zinc chloride and sodium chloride. The condensation of benzylmagnesium chloride and benzene to diphenylmethane can be brought about by small amounts of magnesium and water.
What Is the Structure of Diphenylmethane
Diphenylmethane is an organic compound with the formula (C6H5)2CH2 (often abbreviated CH2Ph2). The compound consists of methane wherein two hydrogen atoms are replaced by two phenyl groups. It is a white solid. Diphenylmethane is a common skeleton in organic chemistry.
Diphenylmethane functions as a reactant in organic synthesis, particularly in the production of dyes and perfumes. It acts as a precursor in the formation of various organic compounds, participating in reactions.
Diphenylmethane′s mechanism of action involves its ability to undergo substitution reactions with other reagents, leading to the formation of new carbon-carbon or carbon-heteroatom bonds. Diphenylmethane can also serve as a building block for the synthesis of complex organic molecules, contributing to the diversification of chemical structures. Its role in these reactions allows for the creation of novel compounds with potential applications in various fields of study.
Safety Precautions for Using Diphenylmethane
Diphenylmethane is a substance that should be handled with caution due to its potential irritant effects on the skin and eyes. Prolonged or repeated exposure to high concentrations of diphenylmethane may lead to skin, eye, and respiratory system irritation. In addition, there is a possibility of central nervous system stimulation and liver damage if it is absorbed into the body.
Therefore, workers should avoid direct skin contact and take measures to minimize their exposure to this substance. It is crucial to prevent fires and explosions during the use and storage of diphenylmethane. The substance is combustible, so it should be stored in a cool, well-ventilated place, away from ignition sources.
It is also important to work in a well-ventilated environment when using diphenylmethane in small amounts, as its irritating odor threshold is low.
Diphenylmethane's safety considerations include avoiding skin contact, preventing prolonged exposure, taking precautions against fires and explosions, working in well-ventilated areas, and complying with safety guidelines provided by the manufacturer and local regulations.
What Is the Importance of Diphenylmethanol
It has uses in perfume and pharmaceutical manufacture. In perfumery it is used as a fixative. In pharmaceutical manufacture it is used in the synthesis of antihistamines / antiallergenic agents and antihypertensive agents . It is used in the synthesis of modafinil and the benzhydryl group is present in the structure of many histamine H1 antagonists like diphenylhydramine.
Benzhydrol is also used in the production of agrochemicals as well as other organic compounds and as a terminating group in polymerizations.
Our Factory
HANGZHOU BETTER CHEMTECH LTD is a comprehensive chemical company specializing in Chemical Production, Management, Application Development and Brand Promotion. Our headquarters is in Hangzhou, China, with service centers in Mumbai, India, and we also have cooperative production bases in China and India.
China's chemical industries are large in scale, but fall short in terms of technical knowhow. In the face of increasingly stringent requirements of the state and the people for the environment and safety production in China, chemical industries are facing large-scale supply shortages. The global chemical supply chain is facing severe challenges in many subdivisions. The chemical industry workforce has lost confidence in the future development of the industry, but BETTER believes that the reconstruction of chemical supply and value chain can lead to the overall development of the industry and the workforce.






FAQ
Hot Tags: diphenylmethane, China diphenylmethane manufacturers, suppliers, factory, allspice flavor, eucalyptus scents, pineapple flavor, jasmine extract, lemon fragrance, cardamom flavor
Previous
No InformationNext
Benzyl AcetateYou Might Also Like
Send Inquiry